Archive for the ‘Discovery’ Category
Sep
14
Posted by jns on
September 14, 2011
I’m still inspired by joining up images from the “Eye for Science” project database with smartphones, and today I implemented another way to make it easy and quick way to turn an image you like into smartphone wallpaper.
All I’ve done is add a QR Code to the image page, i.e., the page you get when you click on the thumbnail image in the widget at the upper right (if you’re looking at this in my blog, and if you’re not you can do so to see what I’m talking about by clicking here).
When you see an image that you think will make a stunning wallpaper image on your smartphone, just use of the available bar-code scanner apps to scan the QR Code, which will translate to the URL of the image page it’s on. You can then easily load the page on your smartphone and use your phone’s image options to make the image your wallpaper. At least, that’s the way it works on my Android phone.
By the way, if you just want to look through some random images you can access this URL : http://scienticity.net/efs. Then, if you see an image you’d like to make into wallpaper, follow the steps above. You can do this repeatedly : every time you access that URL you get a different image randomly chosen from the “Eye for Science” collection. It’s a great way to waste a few minutes or a few hours late at night.
Sep
14
Posted by jns on
September 14, 2011
You may recall my mentioning Scienticity’s “Eye for Science” project, a Flickr group to which members contribute interesting and provocative images that tell a story about science or nature or something related, which images we then try to get in front of others to provide a brief science moment. One way we do this is through a widget that shows a clickable thumbnail of the image; you can see it in action right there on the right of this blog page (or the left, if I’ve redesigned the theme). Every time the page is reloaded, a different image, chosen randomly from the group, shows up. At the time of my writing this the project has been going for a little over two years and the group has 97 members who’ve contributed 994 images. Why not consider joining us?
Ever since smartphones started to appear I’ve wanted to have an “Eye for Science” app that would serve up a random image from the collection whenever the user accessed the app. But, I’ve never taken the time to learn how to program in the necessary way to create such an app. Very recently I upgraded my own phone to something “smart”, an Android model as it turns out, and the thought crossed my mind again, and I made a happy discovery : no programming required!
Well, nearly. Which is to say that I could accomplish the best part of what I wanted with tools that existed on my phone, without writing an app. What I decided I wanted one afternoon was to use an eye-for-science image for my phone’s wallpaper and be able to change it easily whenever the whim arrived to do so, maybe every day (or more often). I did have to do a little behind-the-scenes programming with the Scienticity-hosted webpage that accesses the Flickr database, but that I knew how to do.
So, here’s how I have a pseudo-app–a bookmark, actually–on my Android home page that let’s me view a random sciency image and make it my wallpaper.
I used the “web” app (browser) to access the url, http://scienticity.net/efsm/ ; you can do this in your regular browser, too, there’s nothing magical about it. Accessing this URL returns a random, full-sized image from the Flickr database, along with the title and caption, the same thing you’d get by clicking the thumbnail in the widget on this page.
Once I have this page in my browser I can make a bookmark (“menu / more / add shortcut to home”) on my home screen. From my home screen, then, touching the bookmark opens this URL in my browser with a new random image. If I’m already looking at an image page I can get a new image, randomly selected, by touching “menu / refresh”, because each time I access the URL I get a new random image. Then, I can keep trying images (and enjoying what I see) until I get to one that strikes me as something that will make good wallpaper.
Then — on my phone at least — all I have to do is touch the image on my screen and hold my finger there until I get a menu of options, one of which is “set as wallpaper”. I touch that option and I have new wallpaper!
I’m a little embarrassed to say how inordinately pleased I am to be able to set my wallpaper so easily to new eye-for-science images whenever I feel moved to do so, but there you go.
Please, I invite you to give it a try, It’s quick, it’s easy, and you’ll see interesting things that might make you think “hunh”, which is the goal of the project. Have fun!
Apr
29
Posted by jns on
April 29, 2009

This luminous blob is a “gamma-ray burster”, and exceedingly distant from us: slightly over 13 billion light-years. In fact, it is the current record-holder in the “most distant object seen” category. It was spotted recently by NASA’s Swift spacecraft (about the spacecraft; and about the Swift mission).
Just how a gamma-ray burst happens is still being studied — it’s a big reason behind the Swift mission. GRBs seem to be associated with star remnants collapsing into black holes following a supernova event.
Following the explosion of a big enough star (see “A Star Explodes in Slow Motion“), a shell of matter is expanding around a core that is collapsing into the singularity known as a black hole. In all likelihood this collapsing matter is rotating at very high velocity.
It is thought that lumps of matter falling into the black hole cause some matter to be ejected at very high velocity away from the center in a narrowly collimated jet at relativistic speeds. As this jet passes through the expanding shell of matter around the object, interactions between the jet and the shell of matter produce radiation across a wide spectrum but still in a narrowly visible cone. Sometimes we are fortunate that one of those cones is headed in our direction.
Of this event the NASA press release (“New Gamma-Ray Burst Smashes Cosmic Distance Record“; also source of the image above) says
April 28, 2009: NASA’s Swift satellite and an international team of astronomers have found a gamma-ray burst from a star that died when the universe was only 630 million years old–less than five percent of its present age. The event, dubbed GRB 090423, is the most distant cosmic explosion ever seen. [...]
The burst occurred at 3:55 a.m. EDT on April 23rd. Swift quickly pinpointed the explosion, allowing telescopes on Earth to target the burst before its afterglow faded away. Astronomers working in Chile and the Canary Islands independently measured the explosion’s redshift. It was 8.2, smashing the previous record of 6.7 set by an explosion in September 2008. A redshift of 8.2 corresponds to a distance of 13.035 billion light years.
“We’re seeing the demise of a star — and probably the birth of a black hole — in one of the universe’s earliest stellar generations,” says Derek Fox at Pennsylvania State University [where the flight operations staff is located].
That is incredibly far away!
Sep
26
Posted by jns on
September 26, 2008
Once again Physics News Update delivered what I thought was a really cool story. It concerns an incredibly bright gamma-ray burst from an incredibly distant object–more distant than anything ever seen before: 7,000,000,000 light years away!
The object exploding was an old star that had used up all its fuel, the end of the sequence in which hydrogen is fused into helium, helium is fused into carbon and oxygen, later products fuse into ever heavier elements until the end of the line is reached with lead as a final product in that sequence. When the fusion sequence ends, there is no outward pressure to balance gravity and the star collapses. Its outer shell is blown off, the core can be compressed to a black hole, and the violence can create shock waves of outward expanding gases (that are dispersing those heavy elements around the universe) that interact explosively with each other and give off exceedingly brilliant bursts of electromagnetic radiation — the gamma-ray bursts. For a fantastic description of these last moments in the life of a supernova, you might like to read my posting “A Star Explodes in Slow Motion“.
So brilliant was the end of this start that, as mentioned below, it would have been visible to the naked eye in full sunlight, had you been looking. Fortunately for science, the satellite known as Swift did see it, and had time to let other observers now. And how interesting to remember that this explosion happened 7 billion years ago.
———-
FURTHEST SEEABLE THING.
For the first time in history you could have looked half way back to the origin of the universe with your naked eye. On the night of March 19, 2008 a telescope mounted in space observed a flash from a gamma ray burst, an extremely explosive celestial object, which set several records.
First, if you’d been looking in that direction you would have been able to see, with your own unaided eyes, something at a distance further–seven billion light years–than anything a human being has ever seen in history. Second, since looking out into space is equivalent to looking back in time (it takes the light from distant objects many years to reach the Earth), you would have been witnessing the earliest thing ever seeable by the naked eye.
A new report describes observations made of the explosion by an orbiting telescope called Swift and by some of ground-based telescopes that got in on the action once they were notified by Swift. Swift has three onboard detectors which look not at ordinary visible light but at much more energetic light in the form of x rays and gamma rays. One feature of Swift’s mission is that as soon as it sees something interesting it alerts controllers on the ground so that other telescopes can be turned in that direction. In this way the explosive outburst, whose official name is GRB 080319B, could be tracked by telescopes sensitive to other kinds of light, such as infrared and even radio waves.
The March 19 event is an example of a gamma ray burst. This comes about when certain heavy old stars have used up all their internal fuel. When a star has no more fuel, the force of gravity causes it to contract. If this process is violent enough, the star can blow apart as a supernova. In some special cases, what is left behind is a black hole, and outward going shock waves which, when they criss-cross, can create a brilliant flash of light. For a short time this light is more powerful than that coming from an entire galaxy of stars. The cone of energy flying away from the explosion can be quite narrow, so to be observed from far away, as this object was, it had to be aligned just right to be seen by Swift.
This gamma burst was not the furthest ever observed with a telescope, but it was the brightest in terms of the energy released. So bright, in fact, that it could have been seen unaided in areas of North and South America the night of March 19, if only for about 40 seconds. The splash of light arriving at Swift’s place in orbit that two of Swift’s three detectors were temporarily blinded. Fortunately several telescopes quickly maneuvered into position and could study the stellar explosion as it unfolded. By then the gamma rays, the most energetic part of the light blast, would have died down.
But other types of light continued to issue from the scene. According to Swift scientist Judith Racusin, an astronomer at Penn State, this has become the best-observed gamma ray burst, and the observations have already changed the way we think about bursts work. When you look out at the night sky about 3000 stars are visible. Everything you can see at night is either a planet in our home solar system or one of those stars, all of which are located in our home galaxy, the Milky Way.
The furthest thing you can normally see with the naked eye, and with some difficulty, is the Andromeda Galaxy, about 2.5 million light years away. Only about once a century is a supernova visible from any further galaxy. And by now it’s been 400 years since we’ve seen one of those. That makes GRB 080319B all the more impressive. It breaks the record of most distant seeable-with-the-naked-eye thing by a factor of a thousand.
Located in the Bootes Constellation, the gamma burst is at a distance of 7 billion light years, which means that it took light seven billion years to come from the blast to Earth. That means that a person seeing the visible portion of the blast would have been looking halfway back toward the time of the big bang, when, according to modern cosmology, the universe began. When the blast occurred the sun hadn’t even appeared yet, much less the Earth, much less the human species. (The results appeared [in] Nature magazine, 11 September 2008.)
[Phillip F. Schewe, James Dawson, and Jason S. Bardi, "Physics News Update -- The American Institute of Physics Bulletin of Research News", American Institute of Physics, issue #873, 25 September 2008.]
Sep
24
Posted by jns on
September 24, 2008
The following news arrived in my email recently as a “Physics News Update” item. I thought it interesting enough to share.
———-
CALIFORNIA TREES NOT KEEPING UP WITH CO2.
Forests aren’t absorbing as much carbon dioxide as in the past, and fire suppression might be to blame. Fire suppression in forest encourages the growth of smaller trees and, as a result, significantly reduces a forest’s overall ability to store carbon, according to a new study by scientists at the University of California at Irvine. The researchers, studying forests in California, found that while the number of trees per acre increased in the sixty year period between 1930 and 1990, carbon storage actually declined about 26 percent. This change in the nature of the forests, with greater numbers of smaller trees at the expense of large trees, seems to have been caused by the assiduous suppression of fires by human intervention, the researchers said. Using detailed records, the scientists, compare forests as they were in the 1930s with forests in the 1990s and found that the “stem density” of the forests had increased, which would seem to enhance a forest’s ability to store carbon. In fact, the smaller-tree factor outweighs the denser-forest factor because large trees retain a disproportionate amount of carbon, the researchers concluded.
Climate change, or at least the vast increase in carbon dioxide launched into the atmosphere by the combustion of fossil fuels during the industrial era, has focused scientific attention on the ability of plants, especially trees, to take up and store the added CO2. Trees are not the only carbon sinks (the oceans store vast amounts of CO2), but they are often cited as a key indicator in the fight to stabilize the buildup of greenhouse gases in the atmosphere.
This study, published last month in the journal Geophysical Research Letters, pertains to California only, but Aaron Fellows, one of the study’s authors, believes it will apply to other dry conifer (evergreen) forests in the U.S. western region.
[Phillip F. Schewe, James Dawson, and Jason S. Bardi, "Physics New Update : The American Institute of Physics Bulletin of Research News", #872, American Institute of Physics, 17 September 2008.]
Jun
19
Posted by jns on
June 19, 2008
This news from NASA and the Phoenix Mars Lander seems to be traveling around with near light speed:
Bright Chunks at Phoenix Lander’s Mars Site Must Have Been Ice 06.19.08
TUCSON, Ariz. – Dice-size crumbs of bright material have vanished from inside a trench where they were photographed by NASA’s Phoenix Mars Lander four days ago, convincing scientists that the material was frozen water that vaporized after digging exposed it.
“It must be ice,” said Phoenix Principal Investigator Peter Smith of the University of Arizona, Tucson. “These little clumps completely disappearing over the course of a few days, that is perfect evidence that it’s ice. There had been some question whether the bright material was salt. Salt can’t do that.”
The chunks were left at the bottom of a trench informally called “Dodo-Goldilocks” when Phoenix’s Robotic Arm enlarged that trench on June 15, during the 20th Martian day, or sol, since landing. Several were gone when Phoenix looked at the trench early today, on Sol 24.
[more]
Of course this is big news for the mission and everyone watching it. Finding evidence of water* on Mars–even evidence of water ever having been on Mars–is usually taken as a necessary condition for finding evidence of any life existing, or having existed at one time, on Mars. Whether this is really true is another matter, but water played a necessary role for life to appear on Earth so it’s thought that no water would mean no chance of life.
Now we believe that there is currently frozen water, existing now, today, on Mars. It doesn’t mean extraterrestrial life but a serious road-block (i.e., no water) has been removed. Besides, discovering signs of water, either now or ever, has been a long, long quest for a good number of people, and now they’ve found it.
A lot of people are pretty excited.
__________
* The evidence: four days ago Phoenix was digging a trench when it came upon a solid, white layer. The layer could have been frozen water or maybe something like salt. What’s happened in the last four days? Some small chunks of the white substance, loosened when Phoenix was digging, disappeared. Disappearing means evaporating (more precisely, “sublimating”, which is the word that means evaporating directly from the solid form without first melting), something that salt and other likely substances would emphatically not do, whereas exposed water ice would do exactly that.
Feb
21
Posted by jns on
February 21, 2008

This remarkable image of the Earth rising over the lunar horizon is actually what it seems to be. It is a frame captured from an HDTV video taken on 7 November 2007 by the Japanese KAGUYA spacecraft, which is currently orbiting the Moon on a surveying mission. They tell us that the Earth is seen rising over a spot that is near the south pole of the Moon.
The KAGUYA Image Gallery, the source of this image (click on “HDTV”),* is a delight to look through; every click brings more remarkable sights and insights.
I first learned about the KAGUYA image from this Science@NASA feature for 20 February 2008: “Who’s Orbiting the Moon?“. It gives a nice run-down of all the missions from various countries that already have satellites orbiting the Moon, or will soon.
———-
* I have cropped the image and reduced it substantially so that it will fit in these narrow confines. Visit the website to find the incredibly large, incredibly high-resolution original image.
Nov
01
Posted by jns on
November 1, 2007
While some vaguely scientific notions are passing through my head, here’s a clipping from Physics News. It came as a bit of a surprise to me. I spent most of my laboratory research life doing stuff that came, in one way or another, under the general heading of “thermodynamics”, and yet it never occurred to me to wonder whether we needed to develop a relativistic theory of thermodynamics. Relativistic electrodynamics, sure. Relativistic quantum mechanics, obviously. But relativistic thermodynamics? I guess the worm goes to the bird who thinks of worms first.
What would relativistic thermodynamics be about? The main question would be whether the temperature of some mutually observed object would be measured the same by two different observers, each in a different inertial (i.e., unaccelerated) reference frame. Care to make a guess?
RELATIVISTIC THERMODYNAMICS. Einstein’s special theory of relativity has formulas, called Lorentz transformations, that convert time or distance intervals from a resting frame of reference to a frame zooming by at nearly the speed of light. But how about temperature? That is, if a speeding observer, carrying her thermometer with her, tries to measure the temperature of a gas in a stationary bottle, what temperature will she measure? A new look at this contentious subject suggests that the temperature will be the same as that measured in the rest frame. In other words, moving bodies will not appear hotter or colder. You’d think that such an issue would have been settled decades ago, but this is not the case.
Einstein and Planck thought, at one time, that the speeding thermometer would measure a lower temperature, while others thought the temperature would be higher. One problem is how to define or measure a gas temperature in the first place. James Clerk Maxwell in 1866 enunciated his famous formula predicting that the distribution of gas particle velocities would look like a Gaussian-shaped curve. But how would this curve appear to be for someone flying past? What would the equivalent average gas temperature be to this other observer?
Jorn Dunkel and his colleagues at the Universitat Augsburg (Germany) and the Universidad de Sevilla (Spain) could not exactly make direct measurements (no one has figured out how to maintain a contained gas at relativistic speeds in a terrestrial lab), but they performed extensive simulations of the matter. Dunkel (joern.dunkel@physik.uni-augsburg.de ) says that some astrophysical systems might eventually offer a chance to experimentally judge the issue. In general the effort to marry thermodynamics with special relativity is still at an early stage. It is not exactly known how several thermodynamic parameters change at high speeds. Absolute zero, Dunkel says, will always be absolute zero, even for quickly-moving observers. But producing proper Lorentz transformations for other quantities such as entropy will be trickier to do. (Cubero et al., Physical Review Letters, 26 October 2007; text available to journalists at www.aip.org/physnews/select)
[The American Institute of Physics Bulletin of Physics News, Number 843 October 18, 2007 by Phillip F. Schewe]
Oct
24
Posted by jns on
October 24, 2007
Bubbles seem to be on the net’s mind today. I haven’t kept all the references (here’s one: “Scientists map near-Earth space bubbles“) but it seemed that I kept reading things involving bubbles.
Now, I’ve long been fascinated by bubbles although, despite my being a scientist with a history of doing some hydrodynamics and a relatively keen interest in things dealing with buoyancy, I’ve never worked on bubbles. This is odd, because I’ve long had a question about bubbles that I’ve never answered — probably because I never spent any time thinking about it. Now, apparently, I have to give it some thought.
My question is not particularly well formed, which makes it not a terribly good scientific question.* Nevertheless, I’ve always wondered what it is that determines the bubble size in effervescent drinks. In all classes of drinks with bubbles — soda, beer, champagne, effervescent water — there are those that have smaller bubbles and those that have larger bubbles. As a rule, I tend to prefer smaller bubbles, by the way.
The last time this question crossed my mind and there were people around who might agreeably talk about the issue, we talked about it but came to no conclusion. I think our problem was that we were imagining that it was the effect of bottle size or the shape of a bottle’s neck or some similar incidental phenomenon that determined bottle size, otherwise assuming that all carbonated beverages were otherwise equal. It now seems to me that making that assumption was rather naive and silly.
My thought today is that the bubble size is determined locally in the fluid, that is to say by the physical-chemical environment in the immediate vicinity of the bubble.# It seems to me that there are two questions to consider on our way to the answer:
- Why do bubbles grow?
- Why would a bubble stop growing?
The answer to the second question, of course, might be implicit in the answer to the first.
To begin: what is a bubble? Bubbles happen in mixtures of two or more substances when little pockets of stuff-B collect inside predominantly stuff-B. That means there’s a lot more of stuff-A than stuff-A, which is to say that we usually see bubbles of stuff-B when there’s only a little bit of stuff-B mixed in with a whole bunch of stuff-A.
To be concrete, let’s talk about bubbles of carbon-dioxide (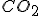 ) in effervescent water, say “San Benedetto” brand.% Now, bubbles have very few features of any physical importance: their sizeis an important one, and the interface between the inside and outside of the bubble, which we might call the surface of the bubble. Some chemical properties of the stuff inside and outside the bubble might matter, too, but let’s save that for a moment.
) in effervescent water, say “San Benedetto” brand.% Now, bubbles have very few features of any physical importance: their sizeis an important one, and the interface between the inside and outside of the bubble, which we might call the surface of the bubble. Some chemical properties of the stuff inside and outside the bubble might matter, too, but let’s save that for a moment.
Now, imagine that we start observing through our clear-glass bottle of San Benedetto water before we take the cap off. The water is clear and there are no bubbles. However, we are aware that there is 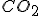 dissolved in the water. In fact, we know that (in some sense) the
dissolved in the water. In fact, we know that (in some sense) the 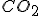 is under pressure because, when we unscrew the cap, we hear a “pffft” sound. Not only that, bubbles instantly appear and float up to the surface of the water, where they explode and make little “plip plip plip” sounds.
is under pressure because, when we unscrew the cap, we hear a “pffft” sound. Not only that, bubbles instantly appear and float up to the surface of the water, where they explode and make little “plip plip plip” sounds.
What makes the bubbles grow? They start out as microscopically tiny things,** gradually grow bigger, until they are big enough to float to the top of the bottle and explode.
To be more precise, bubbles of 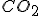 grow if it takes less energy to let a molecule into the bubble across its surface than it costs to keep it outside the bubble. (This is a physicist’s way of looking at the problem, to describe it in terms of the energy costs. A chemist might talk about it differently, but we end up describing the same things.)
grow if it takes less energy to let a molecule into the bubble across its surface than it costs to keep it outside the bubble. (This is a physicist’s way of looking at the problem, to describe it in terms of the energy costs. A chemist might talk about it differently, but we end up describing the same things.)
What causes the bubble to float up? That’s a property called buoyancy, the name of the pseudo-force that makes things float. It’s determined by the relative density of the two substances: less dense stuff floats up, more dense stuff floats down. Notice that the use of “up” and “down” requires that we have gravity around — there is no buoyancy in orbit around the Earth, for instance.
Bubbles, then, will start to float up when they get big enough that they have enough buoyancy to overcome the forces that are holding them in place. That basically means overcoming drag produced by the water’s viscosity, which works kind of like friction on the bubble — but that’s a whole other story, too.
Are we near the answer about bubble size yet? Well, here’s one possibility: bubbles grow until they are big enough to float up to the surface and pop, so maybe they don’t get bigger because they shoot up and explode before they have the chance.
I don’t like that answer for a few reasons. We began by observing that different brand products had different size bubbles, meaning different size bubbles exploding at the surface, so something is going on besides the simple matter of 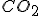 dissolved in water, or else they would all have the same size bubbles. You can look at the bubbles floating up from the bottom and see that they are different sizes in different products to begin with and they don’t change size much on the way up. Thus: bubbles grow very quickly to their final size and their size does not seem limited by how it takes them to float up. Another reason: sometimes bubbles get stuck to the sides of the bottle, but they don’t sit there and grow to arbitrary sizes; instead, they tend to look much like the freely floating bubbles in size.
dissolved in water, or else they would all have the same size bubbles. You can look at the bubbles floating up from the bottom and see that they are different sizes in different products to begin with and they don’t change size much on the way up. Thus: bubbles grow very quickly to their final size and their size does not seem limited by how it takes them to float up. Another reason: sometimes bubbles get stuck to the sides of the bottle, but they don’t sit there and grow to arbitrary sizes; instead, they tend to look much like the freely floating bubbles in size.
There could be a chemical difference, with different substances in the drink limiting the size of the bubbles for some chemical reason. Certainly that’s a possibility, but it’s not what I’m interested in here because I want to explain how there can be bubbles of such obviously different sizes in different brands of what is pretty basically water with insignificant chemical differences in their impurities.
That seems to leave us with one option: the energy balance between the inside and outside of the bubble. Whatever it was that caused it to be favorable for dissolved 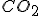 to rush into the bubble at first has some limit. There is some reason that once the bubble reaches a particular size, the energy balance no longer favors
to rush into the bubble at first has some limit. There is some reason that once the bubble reaches a particular size, the energy balance no longer favors 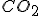 crossing preferentially from water to bubble interior and the bubble stops growing.
crossing preferentially from water to bubble interior and the bubble stops growing.
Now we can look at the “energy balance” matter a little more closely. There are two forces at work. One is osmotic pressure. Osmosis describes how stuff-A moves relative to stuff-B across a barrier (in this case the surface of the bubble); osmotic pressure is the apparent force that causes one substance to move relative to the other across the interface.
The forces at work keeping the bubble in shape are two. One is osmotic pressure inside the bubble, where there is a significantly higher concentration of 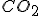 than in the fluid outside, so it creates an outward pressure at the bubble’s surface. The other is the pressure of the water on the surface of the bubble, trying to keep it from expanding.
than in the fluid outside, so it creates an outward pressure at the bubble’s surface. The other is the pressure of the water on the surface of the bubble, trying to keep it from expanding.
So, it looks like the size of the bubbles are determined by a balance between water pressure –determined by the water’s density and gravity — and osmotic pressure inside the bubble, which is caused by the relative concentrations of 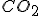 inside and outside the bubble.
inside and outside the bubble.
It would seem, then, that bubbles grow in size until the water pressure on the bubble, which is trying to squeeze it smaller, matches the osmotic pressure of the 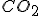 inside the bubble, which is trying to expand the bubble.
inside the bubble, which is trying to expand the bubble.
This works for me, because I know that the osmotic pressure of the 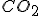 is going to depend on how much was dissolved in the water to begin with. My conclusion, roughly speaking: drinks with bigger bubbles had more carbon-dioxide dissolved in them to start with than drinks with smaller bubbles. At least, that’s my working hypothesis for now, disregarding lots of other possible effects in bubbles. I’ll have to do some experiments to see whether it holds up.
is going to depend on how much was dissolved in the water to begin with. My conclusion, roughly speaking: drinks with bigger bubbles had more carbon-dioxide dissolved in them to start with than drinks with smaller bubbles. At least, that’s my working hypothesis for now, disregarding lots of other possible effects in bubbles. I’ll have to do some experiments to see whether it holds up.
This is just my answer for the moment, my provisional and incomplete understanding. It’s not a subject you can just look up on Wikipedia and be done with. (The articles at about.com that discussed bubbles in sodas I didn’t find credible.) I found several references (one, two, three) that, in journalistic fashion, touted the research of University of Reims’ Gerard Liger-Belair as “Unlocking the Secrets of Champagne Bubbles”, but in fact it was a contribution to nucleation and had nothing to offer about bubble size. The third reference, by the way, has a glaring error in the second sentence, and the rest of the text suggests that the author of the story had little understanding of what was being talked about.
Here, in fact, is a piece about bubbles by Liger-Belair (mentioned in the last paragraph), called “Effervescence in a glass of champagne: A bubble story“. It’s a nice read but it skirts ever so gracefully past the question of bubble size.
As we say in the biz: more research is indicated.
———-
* My usual contention being that most of the work of finding the answer lies in asking a good question.
# What might “immediate vicinity” mean, you ask? In physicist fashion, I’m going to suggest that length-scales in the problem will be roughly determined by the size of the bubbles, so let’s take “immediate vicinity” to mean anything within 1, or 2, or maybe 3 bubble radii. (On closer examination we’d have to consider thermal diffusivity and mass-species diffusivity and such things, but that’s for a more sophisticated analysis.)
%“San Benedetto” is the brand of effervescent water that we prefer here at Björnslottet, in case you were wondering.
**But why! Bubbles first start (“nucleate”) either around small impurities or bits of dust in the fluid, or just from fluctuations in the local concentration of bubble stuff. Let’s leave that as an interesting question for another time and just assume they get started somehow.
Oct
17
Posted by jns on
October 17, 2007
This just in from “Science @ NASA”:
Newly assembled radar images from the Cassini spacecraft are giving researchers their best-ever view of hydrocarbon lakes and seas on the north pole of Saturn’s moon Titan, while a new radar image reveals that Titan’s south pole also has lakes.
Approximately 60 percent of Titan’s north polar region (north of 60o latitude) has been mapped by Cassini’s radar. About 14 percent of the mapped region is covered by what scientists believe are lakes filled with liquid methane and ethane:
The mosaic image was created by stitching together radar images from seven Titan flybys over the last year and a half. At least one of the pictured lakes is larger than Lake Superior.
[excerpt from "New Lakes Discovered on Titan", Science @ NASA, 12 October 2007.]
Isn’t that fascinating: “hydrocarbon lakes” filled with “liquid methane and ethane”!
The photograph accompanying the press release is really quite lovely — it’s what attracted my attention in the first place. Follow the link above to see the photomosaic.


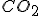 ) in effervescent water, say “San Benedetto” brand.% Now, bubbles have very few features of any physical importance: their sizeis an important one, and the interface between the inside and outside of the bubble, which we might call the surface of the bubble. Some chemical properties of the stuff inside and outside the bubble might matter, too, but let’s save that for a moment.
) in effervescent water, say “San Benedetto” brand.% Now, bubbles have very few features of any physical importance: their sizeis an important one, and the interface between the inside and outside of the bubble, which we might call the surface of the bubble. Some chemical properties of the stuff inside and outside the bubble might matter, too, but let’s save that for a moment.